AI-Translation from the original french article written by Bryan Vaucher, on headstartmd.com
The tech startup model, centered on speed and iteration, collides with the realities of the MedTech sector, where compliance, traceability, and clinical validation are prerequisites for market access. Transposing these methods without adaptation leads to blockages, high remediation costs, and a loss of credibility.
Succeeding in MedTech requires treating design and its documentation as a single, unified product, building a realistic financial timeline, and aligning investors and teams around tangible KPIs. Leaders who adopt this governance manage risk, accelerate valuation, and turn innovation into a scalable, competitive medical device under optimal time and financial conditions.
The GSM Model: Explaining the Priorities of a Tech Startup
The article Software Development in Startup Companies: The Greenfield Startup Model analyzes software development practices in startups, from ideation through to the first beta version. Based on interviews with 13 founders and CTOs, the authors propose the Greenfield Startup Model (GSM), an explanatory model of the dynamics specific to startups.
The findings show that startups operate in an environment characterized by resource scarcity, uncertainty, and intense time pressure. Their central strategy is to accelerate development in order to rapidly deliver a usable product, test product/market fit, and adjust direction based on user feedback.
Figure 1: The Greenfield Startup Model (GSM)
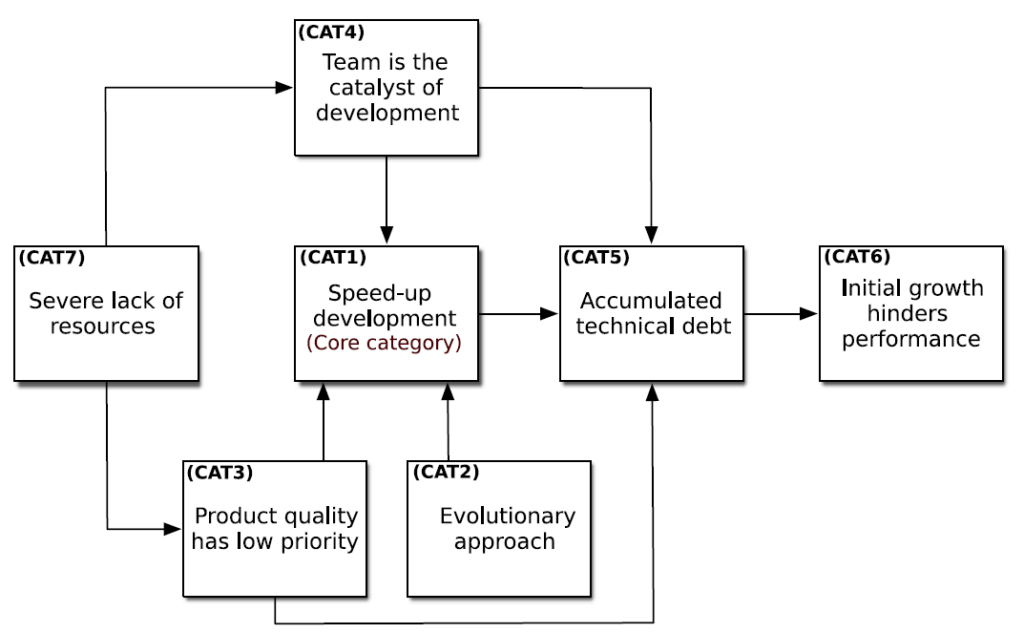
This approach relies on small, highly involved and versatile teams working with flexible processes, informal communication, and minimal documentation. The priority is speed and user experience rather than reliability, robustness, or systematic testing.
This allows for rapid progress, but at the cost of accumulating technical debt due to the absence of thorough architecture, limited use of automated testing, and minimal project management.
These trade-offs work as long as the startup remains small. Growth in users, features, and headcount quickly reveals their limits. Productivity drops and it becomes necessary to restructure the product, pay down technical debt, and introduce more solid practices.
As you can see, this model has both strengths and inherent weaknesses typical of tech startups. On top of these difficulties come fundamental blockers and threats specific to the MedTech sector, which unambiguously demonstrate that operating as a tech startup without adaptation is a dead end.
4 Reasons Why the Tech Model Is Not Viable in MedTech
The following analysis adapts the GSM model to the MedTech context. It highlights the strengths, weaknesses, and threats facing a tech-world startup when it embarks on medical device development, as well as the direct consequences for the organization.
Several major points of tension emerge and considerably amplify the difficulties for a tech startup entering the medical field:
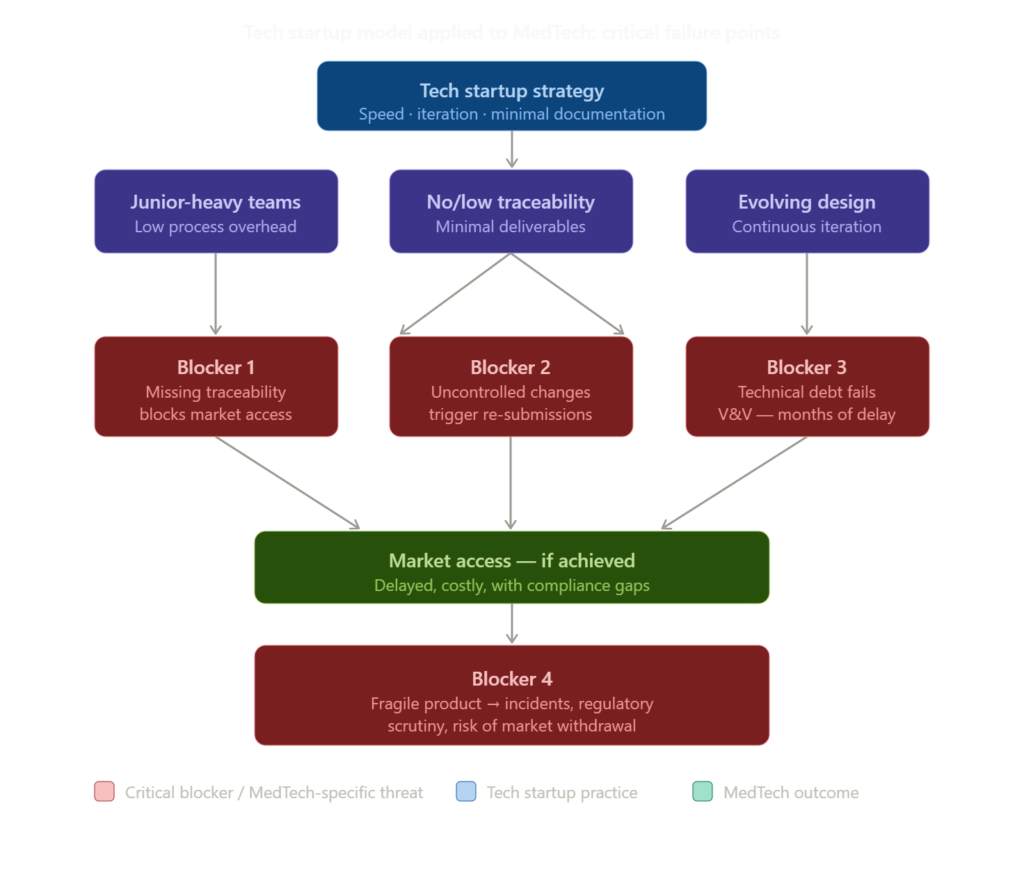
1. Neglecting quality and traceability during design blocks market access. Project deliverables and traceability between requirements, specifications, design, and testing are inseparable from the product itself. Without them, no market launch is possible. The more complex the product, the more costly and time-consuming the remediation.
2. The iterative design approach must be strictly controlled. Any significant modification to a medical device (other than Class I) requires authorization, or even a full new submission. These processes are often accompanied by additional clinical evidence. It is therefore critical to get it right from the initial market launch and to carefully plan future developments.
3. Technical debt strongly increases the risk of failure in verification and validation. An unstable foundation generates unpredictable behavior, especially after modifications. Any failure blocks market access until resolved and can trigger a cascade of delays lasting several months.
4. Even after achieving market access, a fragile product leads to a multiplication of reports and incidents. This results in heightened regulatory scrutiny (FDA, notified bodies), repeated inspections, and the risk of market withdrawal. Managing non-conformities within strict deadlines places a heavy burden on resources, damages the product’s reputation, and hinders clinical adoption.
What Levers Can You Activate for Your Startup?
Managing the Relationship with Investors and Shareholders
An investor who is not specialized in MedTech thinks primarily in terms of return on capital and speed of valuation. The sector’s regulatory and industrial constraints are not spontaneously integrated into their analysis, which requires the CEO to position themselves as a financial risk manager. Promising a fast market launch without genuine experience of the development cycle creates untenable expectations. These inevitably translate into organizational blockages, delays, and a loss of credibility.
Conversely, a lasting relationship of trust is built on realistic, quantified assumptions aligned with market needs.
The first step is to establish a credible financial timeline and clearly show that overly short horizons degrade quality, generate cost overruns, and delay market access.
The real lever of value creation lies in controlled development, where each stage of design and industrialization is carried out with rigor. Non-negotiable timelines must be acknowledged, and the CEO must explain that specifications, risk analyses, and traceability matrices are an integral part of the device. Ignoring them means compromising the success of the project, and for the investor, maximizing the risk of losing the capital they have committed.
Clinical studies and regulatory steps follow the same logic: they must be integrated into planning from the outset and funded to meet their requirements. As long as investors have not validated the resources needed to reach these milestones, any discussion of regulatory timelines remains theoretical. No non-compliant product reaches the market on time, and any attempt to cut corners results in significantly extended delays.
In parallel, pre-market company valuation is built through the regular production of tangible evidence: technical test results, exploratory clinical studies, scientific publications, or performance benchmarks against market standards. These elements reinforce product credibility, consolidate safety and performance data, and support traction with both investors and future users.
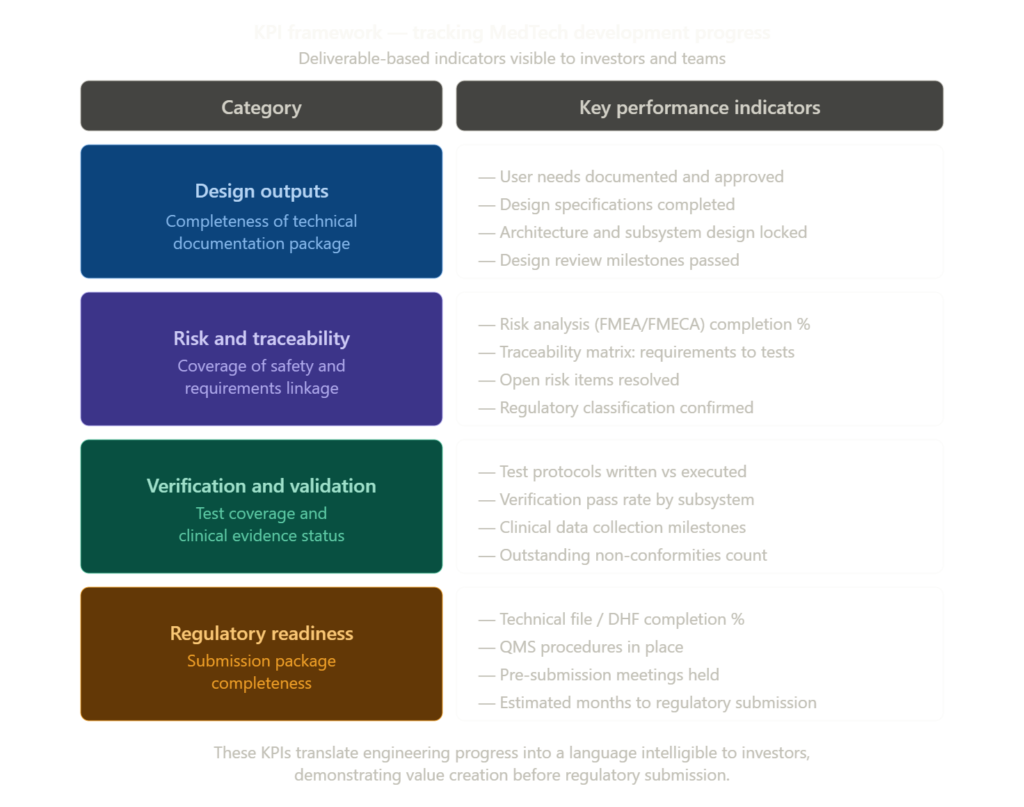
Finally, progress tracking must be objectified through KPIs aligned with the design process. Product deliverables and their completion status become concrete performance indicators, reflecting project advancement in a framework that is intelligible to investors. This reporting approach demonstrates that, even while awaiting regulatory submission, the investment is producing measurable results and that the path to market remains under control.
Managing MedTech Operations with Your Teams
The second critical lever concerns the management of design teams. Many MedTech startups rely primarily on junior profiles, apprentices, or interns, supervised by a CTO or an engineer experienced in general software development.
These profiles are dynamic and cost-effective, but lack the mastery of engineering methodologies required in a sector where traceability, robustness, and a deliverable-driven culture are decisive.
In complex projects combining hardware and software, the standards come from industries such as aeronautics, automotive, or defense, where rigorous project management, traceability tools, a culture of testing, and deliverable-based tracking are the norm.
To succeed in MedTech, it is essential to bring in team members who have already practiced this type of approach. Experienced supervision not only structures the project but also helps develop the skills of junior profiles.
This transition can, however, create tensions: some team members do not embrace this discipline and leave the company. This is a normal stage in an organization’s maturation. What matters is securing key positions, such as a project manager from the medical device world, a software lead with experience in a safety-critical industry, and an organization centered on producing traceable deliverables.
The message to teams must be clear: every technical activity must produce a traceable deliverable. These deliverables are the primary performance indicators for the project and determine market access.
Implementing an appropriate set of traceability tools from the outset (ALM/PLM or equivalent) makes this way of working more reliable and reduces errors. It is the most effective way to align engineering and quality around a common goal: a compliant, scalable product ready for its market.
